CBS News Live
CBS News Bay Area: Local News, Weather & More
Watch CBS News





San Francisco Mayor London Breed announced new legislation that is aimed at cracking down on nighttime drug markets in the Tenderloin.

Logan Webb pitched eight strong innings and extended his scoreless streak to a career-high 19 as the San Francisco Giants beat the New York Mets 5-1 on Tuesday night.

Organizers of the annual Outside Lands Music Festival at San Francisco's Golden Gate Park revealed the lineup for the 2024 event.

The Senate advanced the foreign aid package, which includes a provision that could lead to a ban on TikTok, after months of disagreement in Congress.

The suspects in the killing of a Pleasanton Home Depot employee will see prison time, but his mother's mission for justice is not over yet.

Jurors in former President Donald Trump's trial in New York heard testimony from a former media executive about his efforts to bury negative stories about Trump before the 2016 presidential election.

San Francisco nurses spoke out at the Department of Public Health about urgent issues they say are affecting them at facilities across the city.

Students protesting the violence in Gaza have brought the Mideast conflict home with an encampment occupying the steps of Sproul Plaza on the Cal campus in Berkeley, and they say they're not leaving until their demands are met.

Reverend Cecil Williams, who co-founded Glide Memorial Church and dedicated his life to San Francisco's underserved community, died Monday at the age of 94.

Logan Webb pitched eight strong innings and extended his scoreless streak to a career-high 19 as the San Francisco Giants beat the New York Mets 5-1 on Tuesday night.

Anthony Rizzo snapped a lengthy power drought with a two-run homer that capped a four-run first inning, and the New York Yankees held on for a 4-3 win over the Oakland Athletics.

A controversial figure from the Oakland Athletics' recent history says he wants to be appointed the team's manager when they move to Sacramento.

Michael Conforto homered against the team that drafted him in the first round a decade ago, helping to back rookie Keaton Winn's latest stellar outing as the San Francisco Giants beat the New York Mets 5-2.

Brandon Aiyuk is staying away from the San Francisco 49ers facility at the start of the offseason program as he seeks a lucrative long-term contract.

Watts will raise awareness of serious issues impacting Californians, hold local officials accountable, obtain answers for viewers and provide solutions.

California law says genetic testing companies have to get your permission before they store, use or sell your DNA, but the state itself doesn't have to get your permission and has been storing DNA samples from every baby born there since the '80s. Lawmakers want to change that, but face an uphill battle.

A bill introduced in the wake of our "Handcuffs in Hallways" investigation aims to reduce "unnecessary" calls for police at schools. But one California lawmaker could kill it without a vote.

San Francisco Mayor London Breed announced new legislation that is aimed at cracking down on nighttime drug markets in the Tenderloin.

The suspects in the killing of a Pleasanton Home Depot employee will see prison time, but his mother's mission for justice is not over yet.

Students protesting the violence in Gaza have brought the Mideast conflict home with an encampment occupying the steps of Sproul Plaza on the Cal campus in Berkeley, and they say they're not leaving until their demands are met.

A deadly crash at a bus stop in San Francisco's West Portal neighborhood that killed a family of four last month led city officials to quickly come up with a plan to make the intersection much safer.

Jurors in former President Donald Trump's trial in New York heard testimony from a former media executive about his efforts to bury negative stories about Trump before the 2016 presidential election.

An innovative recycling start-up hopes to make the most of "wish-cycling" -- where people try to recycle things like prescription bottles and ballpoint pens, with no evidence they're ever properly processed.

Bay Area musicians are joining forces with an international alliance to call attention to the impact of climate change.

Students at an elementary school in San Rafael have become experts in the field of recycling, and have lessons for others who need a refresher, including those who work at CBS News Bay Area.

The Arctic fox, a captivating creature with a playful nature and distinctive look is under threat.

More than 100 years ago, wild winter-run Chinook salmon from the icy cold McCloud River ended up in the glacially cold mountain waters of New Zealand, where they continue to thrive today.
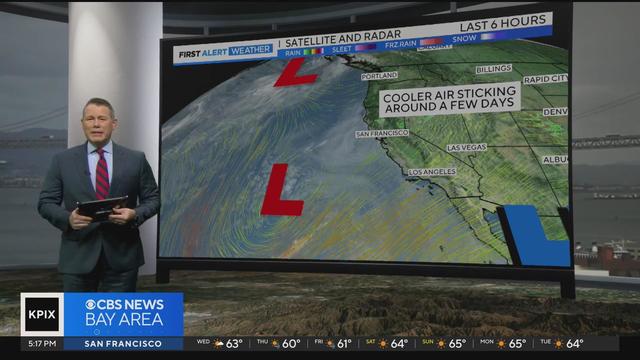
Expect the cooling trend to continue on Wednesday, with cloudy skies inland highs reaching the upper 60s. Showers are possible on Thursday, along with stronger winds. Paul Heggen has the forecast. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv
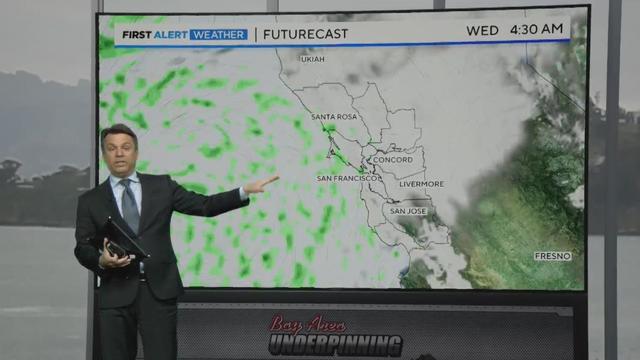
Darren Peck shows us where and when some additional wet weather will be arriving in the Bay Area. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv

Paul Heggen looks ahead to a cooldown over the week with the possible return of rain Thursday.
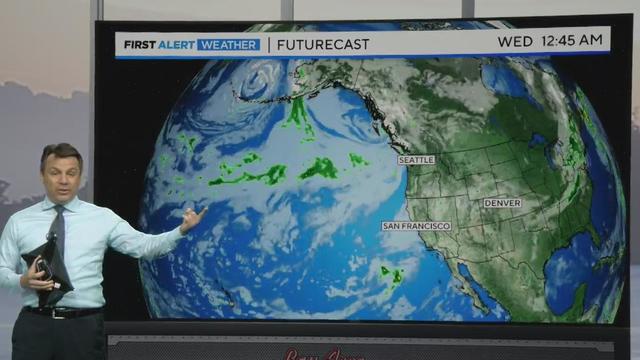
Darren Peck says by the end of the week, our sunny skies will change.

Lauren Toms reports on the latest approach to curb crime in San Francisco's Tenderloin District. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv

The mother of a slain Home Depot security guard and a supporter of Alameda Co. DA Pamela Price’s recall is weighing in on a plea deal reached with the two charged in her son’s murder. Katie Nielsen reports. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv
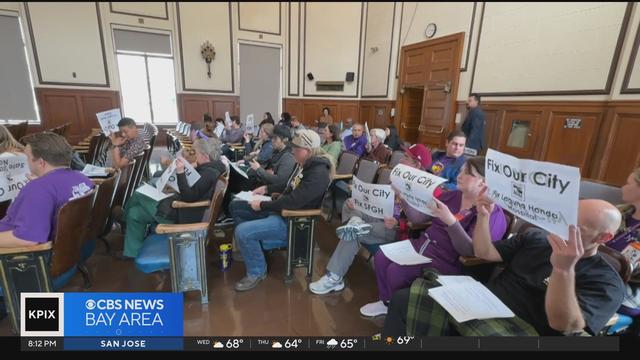
Jose Martinez reports on nurses speaking out at the Department of Public Health about work conditions. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv

Lauren Toms reports on the innovative new prosthetic limb surgery that Air Force veteran Doug Mayo is set to undergo at San Francisco's VA Medical Center. (4/23/2024)

CBS News Bay Area evening headlines for Tuesday, April 23, 2024. Watch full newscasts streamed at the CBS SF website or on the app. Website: http://kpix.com/ YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: https://www.facebook.com/CBSSanFrancisco Instagram: https://www.instagram.com/kpixtv/ Twitter: https://twitter.com/KPIXtv

CBS News Bay Area anchor Ryan Yamamoto asks Dr. Molly Bowdring, psychiatry and behavioral sciences at Stanford Health Care, if people dealing with alcohol addiction should turn to non-alcoholic alternatives, what happens to the brain during addiction, and if non-alcoholic drinks help people with alcohol use disorder drink less

CBS News Bay Area anchor Ryan Yamamoto talks with Dr. Jyothi Tirumalasetty, Allergy & Immunology at Stanford Health Care, about this spring allergy season being worse from last, reasons why, and what we can do to minimize reactions.
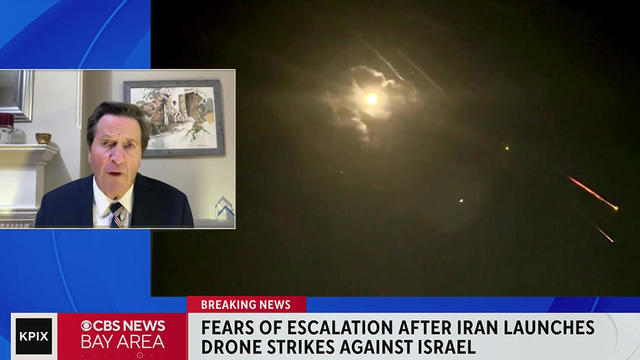
Rep. John Garamendi (D-Walnut Grove) discussed Iran's air attack on Israel Saturday with KPIX anchors Brian Hackney and Andrea Nakano. (4-13-24)

CBS News Bay Area anchors Reed Cowan and Anne Makovec talk to Jill Beyer, OD, optometrist at Stanford Health Care, about how to safely watch a solar eclipse, and the kind of eye damage that can occur without proper protections

Mental illnesses like bipolar disease and schizophrenia can be debilitating, but a new study shows how the keto diet can help. The ketogenic diet is a high-fat, low carb diet. The idea is to help with weight loss by starving the body of carbs for fuel. On the flip side, it could increase bad cholesterol, and potentially raise the risk of heart problems. Dr. Shebani Sethi from Stanford Health Care is in the field of metabolic psychiatry, and CBS News Bay Area anchor Ryan Yamamoto asked her about keto, and how effective it could be when it comes to mental health disorders. "The ketogenic diet is a moderate protein diet, it often gets misconstrued as a high protein diet," said Dr. Sethi. "So it's a moderate protein diet, low carbohydrate, low fat, and it is an intervention that we do use." "Right now we've tested it in those with serious mental illness like schizophrenia and bipolar, and we think it's helpful because it's helping stabilize neuronal membranes, stabilize mood," said Dr. Sethi. "And in the study, what we did is look at a 4-month period to see what the outcome would be for psychiatric symptoms and we improvements in sleep, mood, quality of life, even in some cases reduction in hallucinations frequency and voices that we hear for those with schizophrenia and psychosis," said Dr. Sethi. "So it's been a promising, positive outcome in this pilot study, but there's more research to be do, of course." The study was published in ScienceDirect.

CBS News Bay Area anchor Ryan Yamamoto asks UC Berkeley Haas School of Business professor Olaf Groth, PhD, about the resignation of Boeing CEO Dave Calhoun and what that could mean for the company. Groth also discusses the DOJ's antitrust lawsuit against Apple, and electric vehicle startup Fisker's attempts to avoid bankruptcy

San Francisco Mayor London Breed announced new legislation that is aimed at cracking down on nighttime drug markets in the Tenderloin.

San Francisco nurses spoke out at the Department of Public Health about urgent issues they say are affecting them at facilities across the city.

A deadly crash at a bus stop in San Francisco's West Portal neighborhood that killed a family of four last month led city officials to quickly come up with a plan to make the intersection much safer.

A prisoner left his cell at the jail ward at Zuckerberg San Francisco General Hospital Tuesday morning before being located shortly after, authorities said.

Organizers of the annual Outside Lands Music Festival at San Francisco's Golden Gate Park revealed the lineup for the 2024 event.

The suspects in the killing of a Pleasanton Home Depot employee will see prison time, but his mother's mission for justice is not over yet.

Students protesting the violence in Gaza have brought the Mideast conflict home with an encampment occupying the steps of Sproul Plaza on the Cal campus in Berkeley, and they say they're not leaving until their demands are met.

Anthony Rizzo snapped a lengthy power drought with a two-run homer that capped a four-run first inning, and the New York Yankees held on for a 4-3 win over the Oakland Athletics.

Supporters of the effort to recall Alameda County District Attorney Pamela Price are taking every opportunity to pressure the county Board of Supervisors to call a special election as soon as possible.

Fairfield police are looking for the person responsible for hitting and killing a pedestrian early Tuesday morning.

A caregiver to an elderly South Bay couple has been arrested on elder abuse charges after he allegedly used their debit card to steal thousands of dollars from the couple, deputies said Tuesday.

A volunteer group that picks up trash around San Jose is gaining attention for its high-energy and fast-paced clean
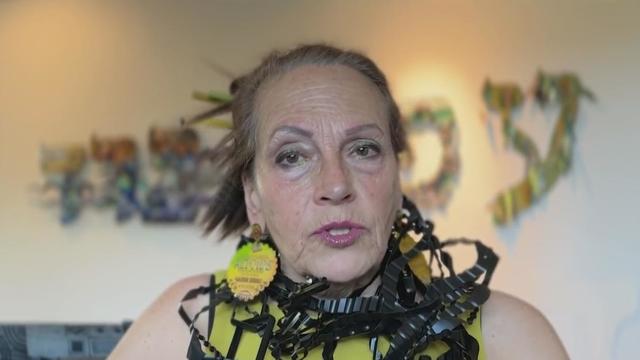
It is often said that "one man's trash is another man's treasure," and for Harriete Estel Berman, she sees beauty in the discarded.

Police in Milpitas arrested a young couple from the Southern California city of Glendale Sunday after they were found with a large quantity of fake jewelry used in scams after a reported theft at a store in the Great Mall.

Firefighters in San Jose responded to a fire at an abandoned building on Saturday morning.

Despite a cyberattack Wednesday, the Solano County Library remains open for patrons.

A helicopter crew airlifted an injured tree trimmer to a Santa Rosa hospital Monday in a dramatic rescue captured on video.

Five people who work as security staff at a Petaluma bar were arrested Saturday night after allegedly beating a patron, police said.

Birds of a feather can gather at the free, family-friendly 15th Annual Point Reyes Birding & Nature Festival at Point Reyes Station Sunday.

A man suspected in a burglary on the west side of Vallejo in March faced additional accusations of rape and kidnapping on Friday, Vallejo police said.

April is National Volunteer Month and organizations like the American Red Cross hope to inspire more people to join their work helping those in need following a disaster or emergency.

Reverend Cecil Williams, who co-founded Glide Memorial Church and dedicated his life to San Francisco's underserved community, died Monday at the age of 94.

Despite being on the front lines of life-and-death situations, being a first responder can be a thankless job, which made one reunion on Wednesday extra special.

Creating change is not easy, but for one museum curator, it's her life mission.

A trio who's led the way in keeping San Mateo County beaches clean is launching a whale of an idea for Earth Day.

San Francisco Mayor London Breed announced new legislation that is aimed at cracking down on nighttime drug markets in the Tenderloin.

Logan Webb pitched eight strong innings and extended his scoreless streak to a career-high 19 as the San Francisco Giants beat the New York Mets 5-1 on Tuesday night.

The suspects in the killing of a Pleasanton Home Depot employee will see prison time, but his mother's mission for justice is not over yet.

San Francisco nurses spoke out at the Department of Public Health about urgent issues they say are affecting them at facilities across the city.

Students protesting the violence in Gaza have brought the Mideast conflict home with an encampment occupying the steps of Sproul Plaza on the Cal campus in Berkeley, and they say they're not leaving until their demands are met.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.

The CDC estimates the U.S. could reach 300 measles cases in 2024 — more than the recent peak two years ago.

California law says genetic testing companies have to get your permission before they store, use or sell your DNA, but the state itself doesn't have to get your permission and has been storing DNA samples from every baby born there since the '80s. Lawmakers want to change that, but face an uphill battle.
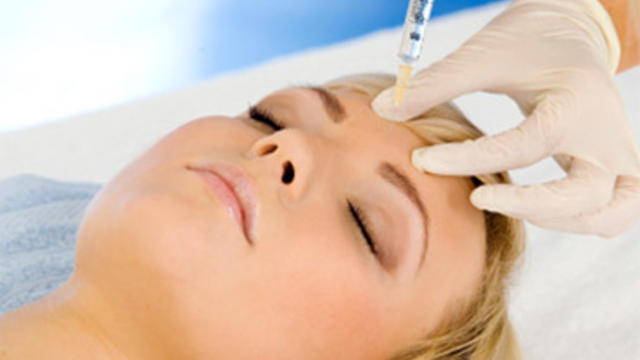
More than 20 people have been stricken after getting fake or mishandled injections in homes and spas, feds warn.

Families of children with a rare autoimmune disorder are hoping new legislation in Sacramento will help their loved ones get the treatment they need.

San Francisco Mayor London Breed announced new legislation that is aimed at cracking down on nighttime drug markets in the Tenderloin.

The suspects in the killing of a Pleasanton Home Depot employee will see prison time, but his mother's mission for justice is not over yet.

A caregiver to an elderly South Bay couple has been arrested on elder abuse charges after he allegedly used their debit card to steal thousands of dollars from the couple, deputies said Tuesday.

Supporters of the effort to recall Alameda County District Attorney Pamela Price are taking every opportunity to pressure the county Board of Supervisors to call a special election as soon as possible.

The defense attorney representing a former Los Angeles-area gang leader accused of killing hip-hop music icon Tupac Shakur in 1996 in Las Vegas said Tuesday his client's accounts of the killing are fiction and prosecutors lack key evidence to obtain a murder conviction.

Ever since the COVID-19 pandemic, the Oakland Unified School District has seen an alarming spike in the number of unhoused students in the school system who deal with a host of challenges far beyond what most children face.

Meteorologist and CBS News Bay Area's resident pilot Lt. Jessica Burch got a treat during Fleet Week, taking to the skies with one of the Blue Angels.

A Bay Area man discovered his devastating loss left him with a new opportunity to rethink how he lives -- follow his journey in virtual reality, 360-degree video.

A groundbreaking medical study involving the UCSF Medical Center has shown some colorectal cancer patients can safely skip radiation treatment and enjoy a potentially higher quality of life.

Every day, San Francisco bar pilot Captain Zach Kellerman goes through what might just be the world's most dangerous commute.

One of San Francisco's oldest LGBTQ bars, on Saturday the Stud re-opened it's doors for the first time in four years at a new location.

A California judge has tentatively sided with state Attorney General Rob Bonta in a dispute over the title of a proposed ballot measure that would require school staff to notify parents if their child asks to change gender identification at schools.

On Friday morning, a special ceremony held on board the USNS Harvey Milk paid homage to the San Francisco gay rights icon who is its namesake.

CBS News national security correspondent David Martin says the naming of the USNS Harvey Milk is a statement that LGBTQ+ rights matter in the U.S.

The naming of the USNS Harvey Milk is just one sign of a very different U.S. military than what many LGBTQ veterans experienced, including one Bay Area army veteran, who reflected on his painful exit from service and how things have changed.

Bay Area pop-punk heroes Green Day played an intimate show at the Fillmore in San Francisco Tuesday for a UN-backed global climate concert.

Bruce Springsteen and the E Street Band played the first of two long-delayed, sold-out concerts at San Francisco's Chase Center Thursday, delivering an epic 29-song set.

Austin-bred band the Black Pumas brought their mix of soul, jazz funk and simmering R&B to the Fox Theatre in Oakland for a packed, sold-out show on Feb. 8.

Riders faced heavy rain and sloppy track conditions at Oracle Park in San Francisco for the Monster Energy AMA Supercross event on Jan. 13.

Iconic electronic pop band Depeche Mode brought their "Memento Mori Tour" to Chase Center Sunday night, playing hits from the last four-plus decades to a full house.

A San Francisco-based nonprofit created by this week's Jefferson Award winner helps low income and unsheltered people stay healthy and feel good about themselves.

A trio who's led the way in keeping San Mateo County beaches clean is launching a whale of an idea for Earth Day.

Two Peninsula mothers are encouraging San Mateo County youth to think about how they can care for the environment and express themselves using the video tools they already use.

An Oakland man is bringing families together to break the cycle of violence in a neighborhood known for violent crime.

A San Francisco woman has spent more than a quarter century helping older adults and people with disabilities remain in their homes safely.

It's hard enough to graduate from one of the most prestigious schools in the country when you're the first in your family to go to college. Imagine doing that while you're also trying to protect your parents from being deported?
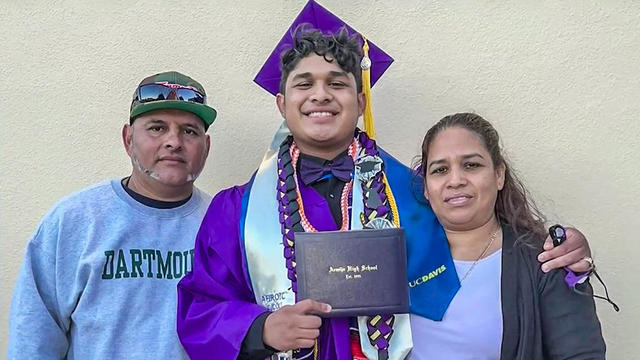
Some students who are the first in their families to go to college face the challenge of balancing a rigorous academic load while still working to help support their family back home.

A onetime pupil has now become a student advisor, giving back after years of mentorship led him to success.

Police departments all over the country are having a hard time finding new officers, but one Bay Area student is criss-crossing the world while preparing for a career in law enforcement here at home.

When most people graduate from college, they tend to focus on one job. But this month's Students Rising Above scholar is currently juggling multiple workplace assignments.