Gilead's Remdesivir Becomes First FDA-Approved Drug For COVID-19 Treatment
The U.S. Food and Drug Administration on Thursday gave the green light to remdesivir as the first approved treatment for patients hospitalized with COVID-19.
Watch CBS News

The U.S. Food and Drug Administration on Thursday gave the green light to remdesivir as the first approved treatment for patients hospitalized with COVID-19.

A Bay Area biotech company says its experimental drug remdesivir improved symptoms when given for five days to moderately ill, hospitalized patients with COVID-19.

Foster City-based Gilead Sciences' experimental drug -- remdesivir -- has been granted FDA approval for emergency use to help coronavirus patients make quicker recovery from infections.

Foster City-based Gilead Sciences announced Wednesday that its experimental drug -- remdesivir -- has proved effective against COVID-19 in a major U.S. government study that put it to a strict test.
When she started at University of California, San Diego, Marlen Canales experienced more than her fair share of first-year college jitters.
The common practice of doctors accepting gifts from drug manufacturers may soon come to a screeching halt in California.
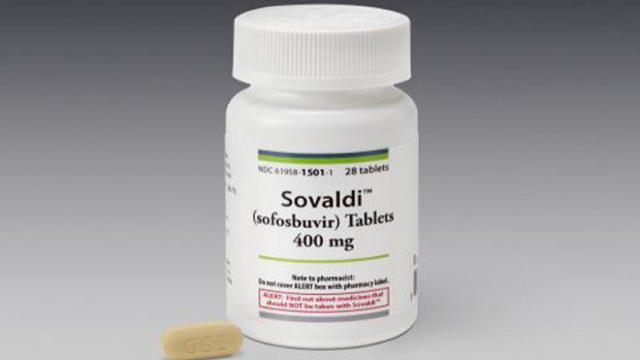
The federal jury in a patent trial has ordered Bay Area-based drugmaker Gilead Sciences to pay Merck $200 million in damages for infringing on patents for hepatitis C drugs.

Drugmaker Gilead Sciences Inc. has halted several patient studies of its cancer drug, Zydelig, because of increased risk of death and serious side effects.

The Massachusetts attorney general's office said Wednesday that it is investigating whether the Bay Area makers of a breakthrough treatment for hepatitis C are violating state law by pricing the drugs too high.

Gilead wants other countries and agencies to back a plan to eradicate hepatitis C by giving away its blockbuster treatment to poor countries.

A Bay Area pharmaceutical firm is among other top drug makers facing scrutiny over enormous profits, the price charged for drugs, and how much the company spends on marketing versus research and development.

An Oregon Medicaid committee is expected Thursday to significantly scale back access to an effective—but expensive—new drug used to treat hepatitis C, the second state to retrict access to the drug in two days.

The maker of a hepatitis C drug that costs about $1,000 per pill on Wednesday delivered a quarterly earnings report that shattered expectations, showing the company tripled its profit which is now in the billions.

Doctors are calling a newly-approved drug a " revolution" in treating the most common blood-borne infection in the United States. But patient advocates warn how those who need it may not be able to afford it; and noisy protests from Paris to San Francisco are trying to drive the displeasure over the price into the open.

Gilead Sciences said Monday that the Food and Drug Administration rejected two marketing applications for HIV treatments, citing quality control problems at the Foster Citry company's manufacturing facilities.
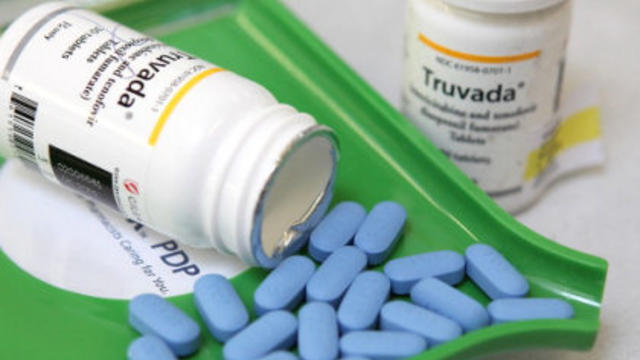
The first drug shown to prevent HIV infection won the endorsement of a panel of federal advisers Thursday, clearing the way for a landmark approval in the 30-year fight against the virus that causes AIDS.

The UC-based California HIV/AIDS Research Program (CHRP) has awarded grants totaling $11.8 million to see if a current HIV drug can be used to prevent the onset of the virus.
California's treasurer on Friday became the second state official to ask leading makers of AIDS medications to extend lower prices and rebates for drugs to help the state's budget crisis.

California's State Controller has asked that pharmacuetical companies lower the price of AIDS medications.
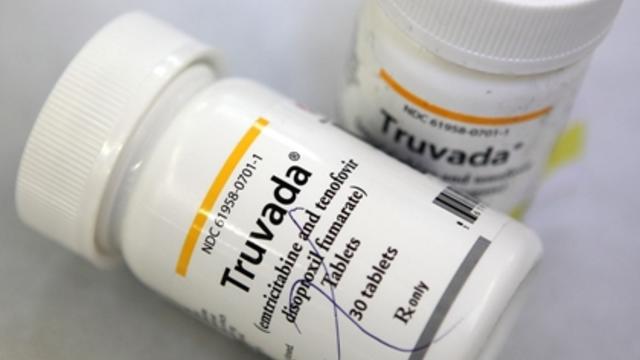
A promising drug shown to decrease the likelihood of transmitting HIV should only be prescribed to men at high risk who are closely monitored by a doctor, federal health officials said Friday.

San Francisco crews knocked down a two-alarm fire in the city's Presidio Heights neighborhood early Friday morning, fire officials said.

An East Bay community is in shock after a solo-vehicle crash Wednesday night killed a family of four.

More than a hundred demonstrators camped on the Cal campus are demanding a ceasefire in Gaza and divestment action by the university.

The Niners ended the first day of the NFL draft by adding a rookie receiver in Florida receiver Ricky Pearsall and keeping its established stars Aiyuk and Deebo Samuel.

The Oakland Athletics beat the New York Yankees 3-1 Thursday night for a four-game split.

San Francisco crews knocked down a two-alarm fire in the city's Presidio Heights neighborhood early Friday morning, fire officials said.

An East Bay community is in shock after a solo-vehicle crash Wednesday night killed a family of four.

More than a hundred demonstrators camped on the Cal campus are demanding a ceasefire in Gaza and divestment action by the university.

In San Francisco, El Farolito soccer club, named after a beloved chain of Bay Area taquerias owned by Salvador Lopez, has flourished.

Bay Area scientists are using cutting-edge technology to better understand the decline in bird populations while finding ways to help species that are challenged.

San Francisco crews knocked down a two-alarm fire in the city's Presidio Heights neighborhood early Friday morning, fire officials said.

In San Francisco, El Farolito soccer club, named after a beloved chain of Bay Area taquerias owned by Salvador Lopez, has flourished.

Mayor London Breed returned from her trip to China last weekend and is now making a visible push to fight for her job as she faces a tough re-election fight with several high-profile challengers.

Parking control officers rallied outside San Francisco Municipal Transportation Agency headquarters Thursday, fearing for their safety as the city ramps up its crackdown on parking violations.

Nearly two weeks after a wedding dress was stolen during a package theft caught on camera in Redwood City, police announced an arrest in the case.

An East Bay community is in shock after a solo-vehicle crash Wednesday night killed a family of four.

More than a hundred demonstrators camped on the Cal campus are demanding a ceasefire in Gaza and divestment action by the university.

Several Bay Area law enforcement agencies collaborated on a months-long retail theft investigation that last week led to the arrest of an Oakland man and netted suspected stolen merchandise worth over $326,000.

Concord police are looking for a man who allegedly grabbed the arm of a middle school student near a parked car on Wednesday.

A horrific single-vehicle crash in Pleasanton Wednesday evening tragically killed a family of four, authorities said Thursday.

Bay Area scientists are using cutting-edge technology to better understand the decline in bird populations while finding ways to help species that are challenged.

A former president of a San Jose elementary school enrichment program allegedly enriched herself by embezzling over $400,000 from the organization, police said Wednesday.

A bomb threat at San Jose City College Wednesday evening led the school to evacuate the campus and cancel all night classes, according to school officials.

The San Jose Sharks announced Wednesday that head coach David Quinn has been relieved of his duties, following one of the worst seasons in team history.

A caregiver to an elderly South Bay couple has been arrested on elder abuse charges after he allegedly used their debit card to steal thousands of dollars from the couple, deputies said Tuesday.

If you're sick of scrolling through streaming services to find a movie to watch on your couch, the North Bay might be your next destination. A Benicia vintage store has set up a 'Free Blockbuster', and it's proving to be popular.

A houseboat went up in flames on Lake Berryessa in Napa County Wednesday, authorities said.

Two drivers were arrested in Santa Rosa after police said a road rage incident between the two led to a shootout Wednesday morning.

Petaluma officers arrested a parolee for allegedly assaulting someone at an apartment complex in the city earlier this week, according to police.

Vallejo police arrested the driver of a stolen car who rammed a patrol car multiple times trying to flee earlier this week, police said.

The Niners ended the first day of the NFL draft by adding a rookie receiver in Florida receiver Ricky Pearsall and keeping its established stars Aiyuk and Deebo Samuel.

The Oakland Athletics beat the New York Yankees 3-1 Thursday night for a four-game split.

The Las Vegas Raiders drafted Georgia's Brock Bowers with the 13th pick of the first round on Thursday, the second year in a row they have taken a tight end high in the NFL draft.

An unprecedented six of the first 12 picks were quarterbacks, an NFL Draft record.

Golden State Warriors point guard Stephen Curry led the league in clutch scoring this season, and was appropriately honored for his late-game heroics Thursday night.

Several Bay Area law enforcement agencies collaborated on a months-long retail theft investigation that last week led to the arrest of an Oakland man and netted suspected stolen merchandise worth over $326,000.

Parking control officers rallied outside San Francisco Municipal Transportation Agency headquarters Thursday, fearing for their safety as the city ramps up its crackdown on parking violations.

Concord police are looking for a man who allegedly grabbed the arm of a middle school student near a parked car on Wednesday.

Nearly two weeks after a wedding dress was stolen during a package theft caught on camera in Redwood City, police announced an arrest in the case.

Two drivers were arrested in Santa Rosa after police said a road rage incident between the two led to a shootout Wednesday morning.

Google's corporate parent Alphabet Inc. has released a quarterly report showing it's still reaping double-digit revenue gains from its digital advertising empire while sowing potentially lucrative new ground in artificial intelligence.

Congo's government is questioning Apple about the tech company's knowledge of "blood minerals" from a conflict zone in the African country that could be smuggled into supply chains.

U.S. regulators are reviving a rescinded rule, laying the groundwork for for a major court fight with the broadband industry.

Documents filed by Tesla with the state of California show nearly 3,000 workers in Fremont and Palo Alto will be among the mass layoffs announced by the electric automaker earlier this month.

Facebook and Instagram parent company Meta says its first-quarter profit more than doubled, boosted by higher advertising revenue and a 6% increase on the average price of ads on its platforms.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.

The CDC estimates the U.S. could reach 300 measles cases in 2024 — more than the recent peak two years ago.

California law says genetic testing companies have to get your permission before they store, use or sell your DNA, but the state itself doesn't have to get your permission and has been storing DNA samples from every baby born there since the '80s. Lawmakers want to change that, but face an uphill battle.
More than 20 people have been stricken after getting fake or mishandled injections in homes and spas, feds warn.

Families of children with a rare autoimmune disorder are hoping new legislation in Sacramento will help their loved ones get the treatment they need.

Mayor London Breed returned from her trip to China last weekend and is now making a visible push to fight for her job as she faces a tough re-election fight with several high-profile challengers.

Mayor London Breed returned from her trip to China last weekend and is now making a visible push to fight for her job as she faces a tough re-election fight. Wilson Walker reports. (4-25-24) Website: http://kpix.com YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: http://www.facebook.com/CBSSanFrancisco Instagram: http://www.instagram.com/KPIXtv Twitter: http://twitter.com/KPIXtv

Parking control officers rallied outside San Francisco Municipal Transportation Agency headquarters Thursday, fearing for their safety as the city ramps up its crackdown on parking violations.

Gavin Newsom was asked to comment about Harvey Weinstein's 2020 rape conviction being overturned on Thursday, and the California governor didn't mince words.

A federal judge has denied former President Donald Trump's request for a new trial in the civil suit brought by the writer E. Jean Carroll, who was awarded an $83.3 million judgment.

One of San Francisco's oldest LGBTQ bars, on Saturday the Stud re-opened it's doors for the first time in four years at a new location.

A California judge has tentatively sided with state Attorney General Rob Bonta in a dispute over the title of a proposed ballot measure that would require school staff to notify parents if their child asks to change gender identification at schools.

On Friday morning, a special ceremony held on board the USNS Harvey Milk paid homage to the San Francisco gay rights icon who is its namesake.

CBS News national security correspondent David Martin says the naming of the USNS Harvey Milk is a statement that LGBTQ+ rights matter in the U.S.

The naming of the USNS Harvey Milk is just one sign of a very different U.S. military than what many LGBTQ veterans experienced, including one Bay Area army veteran, who reflected on his painful exit from service and how things have changed.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Winters Tavern in Pacifica hosts a free garage-rock birthday throwdown Friday night featuring local bands Hot Laundry, the Control Freaks and Minds Without a Face.

The 67th San Francisco International Film Festival begins Wednesday with a variety of screenings including the documentary "Counted Out," which will have its West Coast premiere Sunday.

The defense attorney representing a former Los Angeles-area gang leader accused of killing hip-hop music icon Tupac Shakur in 1996 in Las Vegas said Tuesday his client's accounts of the killing are fiction and prosecutors lack key evidence to obtain a murder conviction.

Organizers of the annual Outside Lands Music Festival at San Francisco's Golden Gate Park revealed the lineup for the 2024 event.

Ever since the COVID-19 pandemic, the Oakland Unified School District has seen an alarming spike in the number of unhoused students in the school system who deal with a host of challenges far beyond what most children face.

Meteorologist and CBS News Bay Area's resident pilot Lt. Jessica Burch got a treat during Fleet Week, taking to the skies with one of the Blue Angels.

A Bay Area man discovered his devastating loss left him with a new opportunity to rethink how he lives -- follow his journey in virtual reality, 360-degree video.

A groundbreaking medical study involving the UCSF Medical Center has shown some colorectal cancer patients can safely skip radiation treatment and enjoy a potentially higher quality of life.

Every day, San Francisco bar pilot Captain Zach Kellerman goes through what might just be the world's most dangerous commute.

An East Bay community is in shock after a solo-vehicle crash Wednesday night killed a family of four. Andrea Nakano and Juliette Goodrich report. (4-25-24) Website: http://kpix.com YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: http://www.facebook.com/CBSSanFrancisco Instagram: http://www.instagram.com/KPIXtv Twitter: http://twitter.com/KPIXtv

The hunts are done with purpose: to leave the community better than they found it.

More than a hundred demonstrators camped on the UC Berkeley campus are demanding a ceasefire in Gaza and divestment action by the university. José Martínez reports. (4-25-24)

CBS News Bay Area evening edition headlines for Thursday Apr. 25 2024. Watch full newscasts streamed at the CBS SF website or on the app. Website: http://kpix.com

Bay Area scientists are using cutting-edge technology to better understand the decline in bird populations while finding ways to help species that are challenged. Len Ramirez and Molly McCrea report. (4-25-24) Website: http://kpix.com YouTube: http://www.youtube.com/CBSSanFrancisco Facebook: http://www.facebook.com/CBSSanFrancisco Instagram: http://www.instagram.com/KPIXtv Twitter: http://twitter.com/KPIXtv

A Redwood City man changed his career path - from running Stanford's Digital Language Lab to going to beauty school - so he could serve his community in a way that's more meaningful to him.

A San Francisco-based nonprofit created by this week's Jefferson Award winner helps low income and unsheltered people stay healthy and feel good about themselves.

A trio who's led the way in keeping San Mateo County beaches clean is launching a whale of an idea for Earth Day.

Two Peninsula mothers are encouraging San Mateo County youth to think about how they can care for the environment and express themselves using the video tools they already use.

An Oakland man is bringing families together to break the cycle of violence in a neighborhood known for violent crime.

It's hard enough to graduate from one of the most prestigious schools in the country when you're the first in your family to go to college. Imagine doing that while you're also trying to protect your parents from being deported?
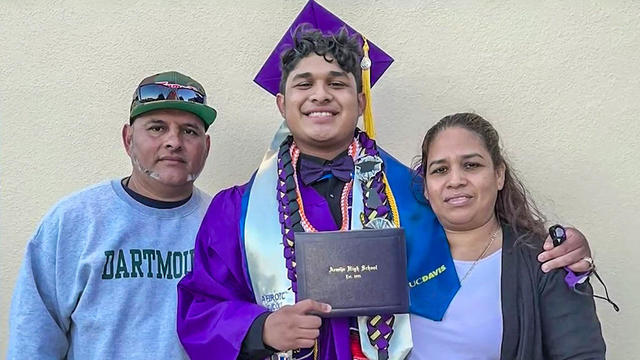
Some students who are the first in their families to go to college face the challenge of balancing a rigorous academic load while still working to help support their family back home.

A onetime pupil has now become a student advisor, giving back after years of mentorship led him to success.

Police departments all over the country are having a hard time finding new officers, but one Bay Area student is criss-crossing the world while preparing for a career in law enforcement here at home.

When most people graduate from college, they tend to focus on one job. But this month's Students Rising Above scholar is currently juggling multiple workplace assignments.